Safe Handling of Radioisotopes
Contents
- Introduction
- Units of Radiation Measurement
- Radiation Quantities and Units
- Radiation Protection Standards
- Table 1 - Fatalities from Accidents in Different Occupations
- Biological Effects of Ionizing Radiation
- Radiation Exposure Limits and Comparisons
- Dosimetry
- Bioassay
- Radiation Physics
- Fundamental Health Physics Principles
- Radioisotope Safety Procedures
- Waste Disposal
- Hazard Evaluations for Commonly Used Radionuclides
- Definitions
- References
Introduction
When used in accordance with instructions, radioactive materials can be used safely in the environment.
The general public is restricted from unnecessary radiation exposure during hydroprobe use, storage, and transportation by virtue of the operating procedures, locked storage, transportation limitations, and legal restrictions imposed by State and Federal regulations.
Operator protection is obtained through training, good gauge design, and following radiological safe work practices (i.e., time, distance, and shielding).
Types of Radiation
Various elements, both naturally occurring (Radium) and reactor produced (Cesium and Americium) are unstable and are slowly decaying to a more stable state. The act of decay produces emissions of energy upon disintegration of the atoms. These emissions are either electromagnetic radiation (gamma rays) or are actual particles (alpha, beta). Other emissions are produced from various radioactive materials; however, we are concerned with only the alpha and gamma radiations and resultant neutrons for purposes of the nuclear soil gauging.
These emissions are detected by appropriate detectors (Geiger Mueller tubes) for gamma rays and (Boron Tri-fluoride or Helium tubes) for neutron measurements. The resultant signals are displayed electronically as an index of soil density and moisture.
All sources are supplied in a sealed stainless steel capsule, doubly encapsulated, and further welded into a stainless steel source rod or located permanently in the gauge housing (see Attachments 1 and 2).
Sources are manufactured by a number of manufacturers' specifications that have been approved by the State of California, Department of Health Services, Radiologic Health Branch.
Sources should never be removed from their mountings and no attempt should ever be made to repair them yourself. Only the manufacturer should perform these source manipulations.
Soil Source Guages
The most common soil gauge sources are:
- Cesium-137 for gamma emission
- Americium-241/Beryllium for neutron emission
- Radium-226/Beryllium for combined gamma and neutron emissions
Gamma Radiation
Gamma radiation is high energy electromagnetic energy capable of penetrating several inches of most material. It is useful for the total mass measurement of heavy materials and is used to determine the total density of soil.
Gamma radiation is emitted in several energy levels by a sealed Radium source or in a single energy level by a Cesium source. The Cesium level is 0.66 million electron volts (MeV) and requires less shielding that the multi-level output of the Radium source. In addition, the fixed spectrum emission is superior for soil density determination purposes.
Gamma sources are relatively easy to shield with dense material like lead, depleted uranium, tungsten, etc.
Neutron Radiation
Neutron radiation consists of small, non-charged particles emitted from the source at an average energy level of 5 MeV. This is known as fast neutron emission. Neutron detectors see only slow, or thermal neutrons; therefore, the fast neutrons must slow down or they will be ignored by the detectors. Neutrons slow down by colliding with other objects (especially light elements like hydrogen) much like a rifle bullet ricocheting from rock to rock.
A simple analogy is that of a golf ball colliding with a bowling ball. The golf ball would rebound with little loss of energy. However, two golf balls colliding would produce a strong loss of energy in each of them, or a transfer of energy from one to the other.
This is what happens when a fast neutron hits a hydrogen atom. The neutron is markedly slowed down. After a few collisions with hydrogen atoms, a fast neutron is reduced to the slow or thermal energy that the moisture detectors in the soil gauge can detect.
Neutron emission occurs when an alpha particle emitter (Americium, Plutonium, or Radium) is mixed with Beryllium powder in a tightly compressed pellet. The alpha particles strike the Beryllium atoms to produce fast neutrons of an average energy of 5 MeV. The suffix Be is attached to an alpha source name to identify the type of neutron source (RaBe, AmBe, PuBe).
Neutron sources are more difficult to shield. Use of hydrogenous moderators may provide shielding but reduces the measuring capacity of the gauge. It is impossible to moderate the neutrons with heavy plastic shielding and still expect the ground moisture to then moderate more neutrons for measurement. Neutron shielding is further complicated in that the thermal neutrons are captured by the moderating material with a resultant emission of gamma radiation of fairly high energy.
Units of Radiation Measurement
Activity (Unit: Curie)
The Curie (Ci) is defined as the activity of that quantity of radioactive material in which the number of disintegrations per second is 3.7E10 (a number nearly the same as the number of disintegrations per second from 1 gram of radium).
Since a Curie is a large amount of radioactivity sub-units of a Curie, a millicurie (mCi, 1E-3 Curie) or microcurie (µCi, 1E-6 Curie), are commonly used to express the amount of activity.
Exposure ( Unit: Roentgen)
The Roentgen (R) is defined as 2.58E-4 coulomb/kg air. This unit is special in that it is defined only for X or gamma radiation in air.
Absorbed Dose (Unit: rad)
The rad is the special unit of absorbed energy. It is defined as that amount of ionizing radiation that deposits 100 ergs/gram of material. For most applications, it can be assumed that
1 Roentgen = 1 rad.
Dose Equivalent (Unit: rem)
The rem is the unit of dose equivalent. The dose equivalent accounts for the difference in biological effectiveness of different types of radiation. It is the product of the absorbed dose (rad) times the quality factor (QF) of the radiation. The QF for x, gamma, and beta radiation is 1, for alpha radiation 20, and varies with energy from 2-11 for neutrons.
Radiation Quantities and Units
Radioactivity: The rate of disintegration per unit time is called activity, which is measured in curies and based on the following standard:
1 curie (Ci) = 3.7 x 1010 dps (disintegrations per seconds)
1 millicurie (mCi) = 3.7 x 107 dps = 1 x 10-3 Ci
1 microcurie (µCi) = 3.7 x 104 dps = 10-6 Ci
1 nanocurie (nCi) = 3.7 x 101 dps = 10-9 Ci
1 picocurie (pCi) = 3.7 x 10-2 dps = 1 x 10-12 Ci
dps = cps/efficiency of detector for specific radionuclide
Radiation Exposure: The measurement of radiation exposure in air as ionizations per unit mass of air due to X or gamma radiation.
1 Roentgen (R) = 2.58 x 10-4 Coulomb/kg air
1 milliroentgen = 2.58 x 10-7 Coulomb/kg air = 1 x 10-3 R
Absorbed Dose: The measurement of radiation absorbed dose (rad) represents the amount of energy deposited per unit mass of absorbing material.
1 rad = 100 ergs/gm = 1 x 10-2 Joule/kg
1 millirad (mrad) = 0.1 ergs/gm= 1 x 10-5 Joule/kg = 1 x 10-3 rad
Dose Equivalent: The measure of biological effect of radiation requires a third unit called a quality factor (QF) which takes into account the different degrees of biological damage produced by equal doses of different types of radiation.
1 rem (Roentgen equivalent man) is the product of the amount of energy absorbed
(rad) times the efficiency of radiation in producing damage (i.e., QF)
1 rem = 1 rad x QF
For X and gamma radiations and most beta, the QF = 1. Therefore, 1 rem = 1 rad
Alpha radiation has a QF of 20
QF for neutrons is 2-11
S.I. System: There is a new system of radiation units, the S.I. system, which is widely used in Europe and gradually being adopted in the United States. The current and new units are shown below with their conversion factor.
| Current Unit | S.I. Units | Conversion |
|---|---|---|
| Curie (Ci) | Becquerel (Bq) | 1 Bq = 1 dps |
| rad | Gray (Gy) | 1 Gy = 100 rad |
| rem | Sievert (Sv) | 1 Sv = 100 rem |
Radiation Protection Standards
Introduction
Radiation protection standards apply to radiation workers or the general population. Standards for the general population are of importance since they serve as a basis for many of the considerations applicable to the siting of nuclear facilities and the design and implementation of environmental surveillance programs. Included in this section are a brief history of the development of radiation protection standards, a review of the goals and objectives sought, and a description of the approach being used to base such standards on the associated risk.
History of the Basis for Dose Limits
Shortly after the discovery of x-rays of 1895 and of naturally occurring radioactive materials in 1896, reports of radiation injury began to appear in the published literature. Recognizing the need for protection, dose limits were informally recommended with the primary initial concern being to avoid direct physical symptoms. As early as 1902, however, it was suggested that radiation exposures might result in delayed effects, such as the development of cancer. This was subsequently confirmed for external sources and, between 1925 and 1930, it became apparent for internally deposited radionuclides when bone cancers were reported among radium dial painters.
With the publication by H.J. Muller in 1955 of the results of his experiments with Drosophila, concern began to be expressed regarding the possibility of genetic effects of radiation exposures in humans. This concern grew and dominated the basis for radiation protection from the end of World War II until about 1960, and led to the first consideration of recommendations for dose limits to the public. With the observances of excess leukemia among the survivors of World War II atomic bombings in Japan, and the failure to observe the previously anticipated genetic effects, however, the radiation protection community gradually shifted to a position in which somatic effects, primarily leukemia, were judged to be the critical (or governing) effects of radiation exposures. This belief continued until about 1970 when it was concluded that, although somatic effects were the dominating effects, the most important such effects were solid tumors (such as cancer of the lung, breast, and thyroid) rather than leukemia. Finally, in 1977 the International Commission on Radiological Protection (ICRP) initiated action to base radiation protection standards on an acceptable level of the associated risk. This effort was provided additional support by the National Council on Radiation Protection and Measurements (NCRP) with the issuance of their updated "Recommendations on Limits for Exposure to Ionizing Radiation" in 1987.
Basic Standards - Philosophy and Objectives
The primary source of recommendations for radiation protection standards within the United States is the National Council on Radiation Protection and Measurements (NCRP). Recommendations of this group are in general agreement and many of them have been given legislative authority through publication of the Code of Federal Regulations by the U.S. Nuclear Regulatory Commission.
- Basic Philosophy
As a general approach, the main purposes in the control of radiation exposures are to ensure that no exposure is unjustified in relation to its benefits or those of any available alternative; that any necessary exposures are kept as low as is reasonably achievable (ALARA); that the doses received do not exceed certain specified limits; and that allowance is made for future developments. - Objectives of the Guides
In general, the objective or goal of radiation protection (and associated standards) is to limit the probability of radiation-induced diseases in exposed persons (somatic effects) and in their progeny (genetic effects) to a degree that is reasonable and acceptable in relation to the benefits of the activities that involve such exposures.
Radiation-induced diseases of concern in radiation protection are classified into two general categories: stochastic effects and non-stochastic effects.- A stochastic effect is defined as one in which the probability of occurrence increases with increasing absorbed dose, but the severity in the affected individuals does not depend on the magnitude of the absorbed dose. A stochastic effect is an all-or-none response as far as individuals are concerned. Cancers (solid malignant tumors and leukemia) and genetic effects are examples of stochastic effects.
- A non-stochastic effect is defined as a somatic effect which increases in severity with increasing absorbed dose in the affected individuals, owing to damage to increasing numbers of cells and tissues. Examples of non-stochastic effects attributable to radiation exposure are lens opacification, blood changes, and decreases in sperm production in the male. Since there is a threshold dose for the production of non-stochastic effects, limits can be set so that these effects can be avoided.
Radiation Protection Standards
- Occupational Dose Limits
Standards provide for an upper boundary effective dose equivalent limit of 50 mSv/year (5 rem/year). On a cumulative basis, however, the newest NCRP recommendations have proposed that the average cumulative effective occupational dose equivalent not exceed 10 mSv (1 rem) times the age of the worker. UC Davis guidelines limit exposure to roughly one-half the state and federal limits. Two key changes or factors to be noted relative to these recommendations are:- The dose limit applies to the sum of the doses received from both external and internal exposures.
- The standards are expressed in terms of the effective dose equivalent, an approach which permits, on a mathematical basis, the summation of partial and whole body exposures.
Dose Limits for the General Population
For a variety of reasons, dose limits for the general population are set lower than those for radiation workers. Justifications for this approach include the following:
- The population includes children who might represent a group of increased risk and who may be exposed for their whole lifetime.
- It was not the decision or choice of the public that they be exposed.
- The population is exposed for their entire lifetime; workers are exposed only during their working lifetime and presumably only while on the job.
- The population in question may receive no direct benefit from the exposure.
- The population is already being exposed to risks in their own occupations; radiation workers are already being exposed to radiation in their jobs.
- The population is not subject to the selection, supervision, and monitoring afforded radiation workers.
- Even when individual exposures are sufficiently low so that the risk to the individual is acceptably small, the sum of these risks (as represented by the total burden arising from somatic and genetic doses) in any population under consideration may justify the effort required to achieve further limitations on exposures.
Concept of Effective Dose Equivalent:
- Basic Objectives:
The objective in developing the concept of the effective dose equivalent was to obtain a system that would provide a unit for radiation protection standards that could be used to express, on an equal risk basis, both whole body and partial body exposures. In developing this approach, the ICRP sought to:- Base the limits on the total risk to all tissues as well as the hereditary detriment in the immediate offspring (first two generations);
- Consider, in the case of internally deposited radionuclides, not only the dose occurring during the year of exposure, but also the committed dose for future years.
Based on a review of data on a world-wide basis (see Table I), the ICRP concluded that, on the average, within a "safe" industry about 100 workers or less would be killed accidentally each year for one million workers employed. Thus, the associated risk of accidental death to the average worker in a "safe" industry would be about:
100/year/1,000,000 = 1E-4/year. - Risks of Death from Radiation Exposures:
Based on epidemiological studies with human populations and biological studies in animals, estimates can be made of the risk of a fatality from cancer or a genetic death for given levels of dose equivalent to various body organs. Some examples are given below to illustrate the thinking that goes into formulation of risk factors:- Studies of the survivors of the atomic bombings in Japan at the close of World War II indicate that for a collective dose of 10,000 person-Sv (1,000,000 person-rem) to the bone marrow, there will be, after latency period, an average of one excess case of leukemia occurring in the population each year. Assuming that each such case ultimately results in a death, and that the excess continues for a period of 20 years, there will be a total of 20 excess cases of leukemia and, therefore, 20 excess deaths due to this exposure. Thus, the risk of death due to leukemia resulting from exposure of the bone marrow can be estimated to be:
20 excess person deaths/10,000 person-Sv = 2-3/Sv - Similar studies among uranium miners have shown that there will be approximately 20 excess cases of lung cancer (and consequently 20 excess deaths, assuming all cases of lung cancer are fatal) for each 10,000 person-Sv (1,000,000 person-rem) to the lungs. Thus the risk of death from lung cancer can be estimated to be:
20 excess deaths/10,000 lung-Sv = 2-3/Sv - For breast cancer, epidemiological data have shown that there is an excess of about 100 breast cancers per 10,000 person-Sv (1,000,000 person-rem) to the female breasts. Assuming that breast cancer is fatal 50% of the time; and assuming that the population being exposed consists of 50% men and 50% women, then the risk of excess deaths due to exposure to the female breasts can be estimated to be:
100 excess cancers/10,000 breast-Sv x (0.5 fatality rate) x (0.5 of population being female)= 2.5-3 / Sv - For thyroid cancer, epidemiological data have shown that there is an excess of about 100 thyroid cancers per 10,000 Sv (1,000,000 rem) to the thyroids in humans. However, the fatality rate for thyroid cancer is only about 5%, so the risk of death due to cancer of the thyroid resulting from exposure to ionizing radiation is:
100 excess cancers/10,000 thyroid-Sv x (0.05 fatality rate)=5-4/Sv
- Studies of the survivors of the atomic bombings in Japan at the close of World War II indicate that for a collective dose of 10,000 person-Sv (1,000,000 person-rem) to the bone marrow, there will be, after latency period, an average of one excess case of leukemia occurring in the population each year. Assuming that each such case ultimately results in a death, and that the excess continues for a period of 20 years, there will be a total of 20 excess cases of leukemia and, therefore, 20 excess deaths due to this exposure. Thus, the risk of death due to leukemia resulting from exposure of the bone marrow can be estimated to be:
Similar calculations can be made to estimate the excess deaths due to exposures of other body organs, as well as genetic deaths due to exposure of the reproductive organs.
Table 1 - Fatalities from Accidents in Different Occupations
Fatalities From Accidents in Different Occupations (x 10,000 Per Year)
| Category | Occupation | Fatalities Per Year |
| Safe | Trade | 0.5 |
| Safe | Manufacturing | 0.6 |
| Safe | Service | 0.7 |
| Safe | Government | 0.9 |
| Less Safe | Transportation & Utilities | 2.7 |
| Less Safe | Construction | 3.9 |
| Less Safe | Agriculture | 4.6 |
| Less Safe | Mining, Quarrying | 6.0 |
| Least Safe | Sports | 15 |
| Least Safe | Deep Sea Fishing | 30 |
| Least Safe | High-rise Steelworkers | 50 |
| Least Safe | Farm Machinery Workers | 80 |
Biological Effects Of Ionizing Radiation
Physical and Chemical Effects of Ionizing Radiation
- Ionizing radiation is so named because its initial interaction with matter is the ejection of an orbital electron from an atom, forming a pair of ions with opposite charges. Radiation passing through living cells will ionize or excite atoms and molecules in the cell structure. This produces ions and radicals within the cell (mostly from water molecules). When these radicals and ions interact with other cell materials, damage can result. Certain levels of cellular damage can be repaired by the cell. Further levels can result in cell death.
- May directly involve and damage biologically important molecules in the cell - Direct Effects. Damage to the DNA molecule or a chemical change in other cellular material are the primary results. Damage to the DNA molecule can result in somatic mutations that may show up years after the exposure or genetic mutations that require several life spans to appear.
- May initiate a chain of chemical reactions, mediated through cellular water, leading to ultimate biologic damage - Indirect Effects. An hydroxyl poisoning effect on the cell membrane results in a change in its permeability. Inactivation and release of enzymes is the primary result.
- The unit of radiation dose is the rad which equals 100 ergs of energy absorbed per gram of tissue.
- Biological effects of all types of ionizing radiations are similar. Some radiations are more efficient than others, however, and produce more biological damage per rad dose.
- The rem is the unit of biological dose equivalence which takes into consideration the differing efficiencies of the different radiations.
- The Dose Equivalence in rems is obtained by multiplying the dose in rads by the Quality Factor (QF) of the particular radiation. The QF is related to its ionization density.
QF = 1 for x-rays, gammas, and beta particles
QF = 2 - 11 for neutrons
QF = 20 for alpha particles
Cellular Effects of Ionizing Radiation
- Cell killing is responsible for acute somatic effects of radiation. It occurs by two mechanisms:
- Inhibition of mitosis which results from moderate doses and leads to delayed cell death.
- Immediate cellular death which results from very high doses.
- Alteration of cellular genetic material consistent with continued cell proliferation: Usually manifests no visible change in cellular appearance but a point (recessive) mutation is formed, which may or may not be passed to future generations.
Systemic Biological Effects of Ionizing Radiation
- Somatic effects:
Abnormality may become manifest only after many generations of cell replication: proposed mechanism for long-term somatic effects of radiation - carcinogenesis, nonspecific life shortening. (These are non-stochastic effects.) - Genetic effects:
If involves gonadal cells, mutations are passed on to offspring. Increase in number of "recessive" mutations in population pool leads to increased probability of abnormalities in offspring due to chance mating of individuals carrying same mutation. (These are stochastic effects.)
Acute Somatic Effects of Radiation Exposure in Humans
- Related to killing of cells, generally in tissues where cells are rapidly proliferating. Observed effects usually occur 1-3 weeks after radiation exposure.
- Systems of primary involvement:
- Hematopoietic system
Chief organ: bone marrow
Symptom latency: days to weeks
Death threshold: less than 500 rem
Characteristic symptoms: Malaise, fever, fatigue, infection, hemorrhage, and anemia. Low counts of platelets, lymphocytes and erythrocytes result in low resistance to infection and a decreased clotting ability. - Gastrointestinal system
Chief organ: small intestine
Symptom latency: hours to days
Death threshold: 500-2000 rem
Characteristic symptoms: Malaise, nausea, vomiting, diarrhea, fever, dehydration, G.I. malfunction, and electrolyte loss. The intestinal epithelium is destroyed. - Central nervous system
Chief organ: brain
Symptom latency: minutes to hours
Death threshold: 2000-5000 rem
Characteristic symptoms: lethargy, tremors convulsions, encephalitis, meningitis, and edema. Acute inflammation and vascular damage results in neuronal functional impairment.
- Hematopoietic system
Dose relationships
- 0-150 rem - none to minimal symptoms. Perhaps long-term effects many years later.
- 150-400 rem - moderate to severe illness due to hematopoietic derangement.
- 400-800 rem - severe illness. LD50 in man probably about 500 rem. GI damage at higher doses.
- Above 800 rem - 100% fatal, even with best available treatment.
Partial body exposure
Effects depend on particular tissue or organ exposed, but significant acute changes are usually seen only after a fairly high radiation dose (>1000 rem).
Long-Term Effects of Exposure to Ionizing Radiation
- General characteristics: Usually occur many years after acute or chronic radiation exposure.
- Biologic Effects of Ionizing Radiation:
- Occur with much lower doses and dose-rates: insufficient to cause acute somatic effects.
- Probably related to irreparable damage to genetic material in cells which are capable of continued cell division.
Radiation Carcinogenesis in Humans
Genetic and proliferative alterations of cells require years to many lifetimes to develop.
- Tumor development: Ionizing radiation in large amounts is an effective carcinogenic agent.
- Sterility: Temporary sterility can be induced at exposure levels of approximately 150 rem. Females are more often permanently affected than males.
- Cataracts: Due to the high sensitivity of the lens of the eye, opaque areas of the lens develop after exposure of 200-600 rem.
- Life-shortening: The aging process is increased. Nutrition to the cell appears to be impaired. The total cell number is decreased and there is a modification of the composition of cellular material.
- Fetal damage: The fetus is highly radiosensitive due to the rapid division of cells. No measurable fetal damage has been seen at exposures less than one rem.
- Chromosomal damage: Detection of chromosomal damage requires many generations. An Oak Ridge study suggests that low intensity (1-10 rem/day) continuous exposure has only 1/4 - 1/10 the mutagenic efficiency of acute exposures.
Law of Bergonie and Tribondeau
Radiation sensitivity of cells generally varies directly with the rate of proliferation and the number of future divisions, and inversely with the degree of morphological and functional differentiation.
The following is listed from least to most radiosensitive:
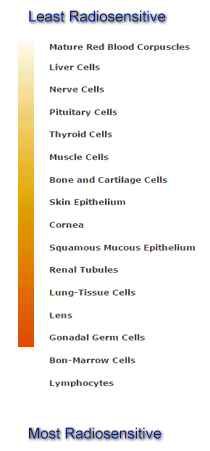
Factors that Influence the Severity of Absorbed Dose
- Internal Radiation
- Amount of Radioactivity
- Radioisotope
- Nature or type of the emission
- Critical Organ
- Physical half-life
- Biological half-life
- Age, weight, sex
- External Radiation
- Amount of Radioactivity
- Nature or type of the emission
- Radiation Energy
- Time
- Distance
- Shielding
- Age, weight, sex
- Area of the Body Exposed
Radiation Exposure Limits and Comparisons
Dose Equivalent Limits (Monitored Radiation Workers)
| Targe Tissue | Regulatory Limit | UC Davis Guideline |
| Whole Body | 5000 mrem/year | 2500 mrem/year |
| Extremities | 50000 mrem/year | 25000 mrem/year |
| Skin of the Whole body | 50000 mrem/year | 25000 mrem/year |
| Fetus | 500 mrem/gestational period | 50 mrem/month |
Common Radiation Exposures (Natural Sources and Human Made)
| One Coast to Coast Flight | 3 mrem |
| Natural Background Radiation in the U.S. | 150 - 300 mrem/year |
| Chest Radiograph, A/P view | 2 mrem/view |
| Chest Radiograph, Lateral view | 10 mrem/view |
| Screening Mammography | 40 mrem/view |
| Computerized Tomography of Body | 1000 mrem |
Biologically Significant Radiation Exposures (Absorbed/Acute Exposure)
| Risk of contracting cancer increased 0.05% | 1000 mrem |
| Temporary blood count change | 50000 mrem |
| Permanent sterilization in men | 350000 mrem |
| Permanent sterilization in women | 250000 mrem |
| Transient Skin Erythema | 200000 mrem |
| Cataract formation | 50000 mrem |
Dosimetry
Dosimeters are devices that quantitate the amount of radiation to which a person has been exposed.
Types of Dosimetry Used on Campus
- Film Dosimeters
The dosimeter used most often on campus is the film badge, comprised of one of two small x-ray films enclosed within a light-tight envelope and plastic holder. The badge is worn from one to four weeks on the trunk of the body, usually at waist level or on the collar. Photographic film in the form of thin, even layers of emulsion is spread on a thick paper support base. The emulsion consists of small silver halide crystals embedded in a gelatin matrix. When the badge is exposed to radiation, energy is transferred to the emulsion causing silver ions to cluster together. These silver clumps are called latent image centers. This film detects x and gamma rays, beta particles greater than 1 MeV, and neutron radiation, except fast neutrons. Fast neutrons require a separate type of film. the amount of exposure is related to the length of the track that is left on the film. The accuracy of film badges is plus or minus 10 mrem.
There are two types of film badges. One badge monitors x, gamma and beta radiation. The other contains an additional film which is sensitive to neutrons. Each holder has filters on the from and back sides containing an open window, then plastic aluminum, cadmium, and lead filters. The type of radiation (i.e., x, gamma, beta, or neutron) can be determined by observing the relative darkening of the film behind each filter.] - Thermoluminescent Dosimeters (Whole Body Exposure Monitors)
In some situations, thermoluminescent dosimeters containing lithium fluoride or calcium fluoride chip and powder cartridges are used in place of x-ray film as a personnel monitor. Exposure of these materials to ionizing radiation results in the trapping of electrons in energy levels above those occupied normally. When the dosimeter is heated, these electrons are liberated from the traps. As the electrons return to their normal levels, visible light is released. The amount of light released is measured and is proportional to the exposure of the dosimeter to radiation. These materials are x, beta, and gamma sensitive and exposure is reported as being either deep and/or shallow energy penetration. - Finger Ring Dosimeters
To monitor hand exposure to radioactive materials thermoluminescent dosimeters in the form of finger rings are worn on the dominant hand with the TLD chip facing the source of radiation. The TLD process is described above. It important is assure chip placement in the dosimeter prior to each use.
The dosimetry reporting company, an independent contractor, will report exposures per individual for the finger ring and deep and/or shallow energy penetration for the whole body.
Precautions on Use of Dosimetry
When not in use, store your dosimetry in an area free of ionizing radiation. If you lose, contaminate, get your badge or rings wet or leave them in the sun for an extended period of time, please notify the campus Office of Environmental Health and Safety, Health Physics or the UCDMC Health Physics Office. While wearing a lead apron, place your badge outside the apron.
- Film badges
Fading of the latent image centers is produced with time, high humidity, and high temperature. - TLD
The lithium fluoride chips and powder are highly sensitive to heat and moisture.
Distribution and Use of Film Badges
- Dosimetry is issued by the campus Office of Environmental Health and Safety, Health Physics (530-752-1493) or the UCDMC Health Physics Office (916-734-3355) based on procedures used and the type of equipment used.
- Badges may be exchanged weekly, monthly, or quarterly, depending upon the type of equipment or type and amount of materials used and experimental design.
- Both the campus and UCDMC Health Physics sections document the dosimetry readings for you and the State of California, Department of Health Services, Radiologic Health Branch.
Dosimetry Records
All dosimetry records are on file with the Office of Environmental Health and Safety, Health Physics. Upon your request, EH&S will supply you with your dosimetry history. If at any time your exposure exceeds the campus guidelines or is unusually high, a health physics staff member will notify you of the incident.
Bioassay
Thyroid Scans
A NaI (Tl) detector placed flush with an individual's thyroid is used to detect any uptakes of radioactive iodine (123I, 125I, or 131I) in experimenters using a volatile form or process of any radioiodine. For persons using greater or equal to 10 millicuries, this scan will be done after each use within 24-72 hours.
Urinanalysis
Urine samples are counted in a liquid scintillation counter to detect the uptake of tritium in individuals using a volatile form or process of tritium greater than 10 millicuries on the open laboratory bench and 100 millicuries in a fume hood.
Whole Body Scans
A person is placed in a whole body counter and a count is performed using detectors for gamma and bremsstrahlug radiation for a whole body count. Determination of internal exposures is calculated from these bioassay methods. The intake limits are established by the NCRP. NOTE: The individual's radiation use authorization (RUA) will contain specific information regarding the bioassay requirements.
Radiation Physics
Decay
Radioactive atoms decay in accordance with the relationship:
N = Noe-lambda * t - Where N is the number of atoms remaining from the initial number (No) after an elapsed time of t. The decay constant lambda is specific for the particular radionuclide and has units of time-1.
lambda = ln2 / T 1/2 - The decay equation can also be used to describe the activity (A) at any time (t) folloing the initial activity (Ao) by the following relationship:
A = Aoe-lambda * t - Activity levels are usually expressed in curie, millicurie, or microcurie units or in disintegrations per minute or per second. There is a relation between the activity A and the number of atoms N.
A = lambda * N - where lambda is the decay constant for the particular radionuclide. The half-life (T1/2) of a radionuclide is the time required for the activity (A) or number of atoms (N) to be reduced by decay to half of the initial number. Half-lives are specific for the different radionuclides and are related to the decay constant by the following relationship:
T1/2 = ln2 / lambda = 0.693/lambda - Substitutions of the half-life for decays constants give the relationship:
N = Noe-(ln2/T1/2)(t) and A = Aoe-(ln2/T1/2)(t)- These equations can be used to calculate either the number of atoms or activities at times following or previous to a known number of atoms or activity.
Calculating Radiation Doses
A. Beta Radiation
The dose rate (rads/hour) at 1 centimeter from a beta particle point source (neglecting absorption) can be approximated by:
Dose Rate 1 cm = 200 x mCi
This approximation can only be used if the distance from the point source is well within the range of the beta particle.
B. X and Gamma Radiation
The exposure rate (mR/hr) at 1 foot from a X or gamma ray point source can be approximated by:
Exposure Rate 1 ft. = 6CEn
where:
C= activity of source in mCi
E = photon energy in MeV
n = abundance of photon
Fundamental Health Physics Principles
Factors in Maintaining ALARA
The ALARA concept in radiation protection is to keep your radiation exposure as low as reasonably achievable. You can limit your exposure to radiation by using the three methods of (1) time, (2) distance, and (3) shielding.
Time
Reducing the time of exposure is a very practical method of radiation protection. The longer the time exposed to a radiation field, the greater the total exposure. The standards for permissible levels of radiation in unrestricted areas and how time influences them is outlined below.
- Radiation levels which, if an individual were continuously present in the area, could result in a dose in excess of two millrems in any one hour; or
- When an exposure rate exceeds 2 millirem per hour, calculations must be made to determine how long an individual can remain in the area.
Distance
Distance is a very effective shielding measure and often the least expensive means of radiation protection. As one moves away from the source of radiation the amount of radiation at a given distance from the source is inversely proportional to the square of the distance (inverse square law).
|
ID2 = id2I = intensity at a distance (D) from a point source |
Shielding
Shielding is also a practical means of radiation protection. For alpha and beta radiation, very little shielding is required to absorb the emissions completely.
- alpha stopped by paper
- beta stopped by one-inch wood, one-quarter inch plexiglass
- x-rays and gamma rays attenuated by concrete or lead
- neutron - hydrogen rich materials
In general, as the density and/or thickness of a shielding material increases, the absorption of radiation emissions by the material also increases. Usually, the higher the atomic number of the shielding material, the higher its density.
Radiation Shielding
Half value layer - the thickness of a material that decreases the x-ray beam intensity by one-half.
| Energy (keV) | Air (cm) | Water (cm) | Al (cm) | Lead (cm) |
| 10 | 115 | 0.1 | 0.01 | 5E-04 |
| 20 | 765 | 0.9 | 0.07 | 7E-04 |
| 30 | 1648 | 1.9 | 0.22 | 2E-03 |
| 40 | 2319 | 2.6 | 0.43 | 4E-03 |
| 50 | 2766 | 3.1 | 0.67 | 8E-03 |
Radioisotope Safety Procedures
These procedures have been developed through investigation of contamination and exposure incidents and monitoring of high activity experiments. Experience has shown that there is considerable reduction in laboratory contamination and personnel exposure when these basic precautions are used.
Work Area
- Open Bench
If possible, locate a radioisotope work area away from heavy traffic and doorways. This will reduce the severity of spreading contamination in the event of a spill, and will also reduce interruptions of delicate, hazardous operations. Clear ample bench area of unnecessary items and cover, using an absorbent material with impervious backing. Keep all equipment associated with the radioactive materials in this defined space and allow no other use of it until the experiment and the monitoring is completed. The covering should then be changed. Label all radioisotope containers and contaminated equipment and keep them well to the rear of the laboratory bench. Large volumes of radioactive liquid that could not be contained by the bench covering must be kept in trays or other secondary container.
Plastic liners for waste containers, labeling tape, absorbent paper, and disposable gloves are available from the Central Storehouse. Room labels and labels for waste containers are available from the Office of Environmental Health and Safety. - Enclosures
If the radioactive material presents an airborne hazard potential from dust or vapor, some containment will be required. Partial containment is offered by chemical fume hoods. Glove boxes provide additional protection. Please call EH&S (2-1493) for a hood inspection before beginning use of radioisotopes in any type of hood.
Preparation
- Equipment
Assemble all items necessary for the procedure, so that delays during the experiment can be avoided. Hazardous operations should not be left unattended while additional equipment is located and prepared. Run through the procedure mentally, making a list of all materials that might possibly be needed. - Emergency Supplies
Attempt to foresee what problems might occur, including spills, and store the appropriate materials close to the work area. Some examples would be extra gloves and absorbent covering, wipes, plastic bags, forceps, decontamination solution. If a spill can be contained immediately, extensive contamination is less likely. - Protective Clothing
Laboratory coats and gloves are required for all handling of unsealed radioactive materials. Double gloves are recommended and should be changed frequently in order to limit the spread of contamination. If a laboratory coat becomes contaminated, change it. Information on more specialized protective equipment is available from EH&S, Health Physics. - Dosimetry - Instrumentation
EH&S assigned Dosimetry (TLD or film badges) must be worn as required (see "Dosimetry" above). Portable survey meters counters are required to monitor work with all radioactive materials except 3H (tritium). Make sure the instrument is operable and place it in the immediate work area (see "Self-Monitoring"). - Radioactive Wastes
Place adequate waste receptacles near the work surface so waste may be contained immediately after it is produced. For dry waste, a plastic bag on the work bench is advised. This avoids spreading contamination between the work bench and waste box during the procedure. A bag hung at the bench front is not advised due to possible exposure problems and the potential rupture and floor contamination. The approved EH&S liquid waste containers (5 gallon jerry jugs) should be placed in a tray to prevent contamination spread during filling and to contain the waste if the container ruptures. Shield the waste receptacles as necessary.
Handling Procedures
- Shipment Ordering
Usually the highest activity is handled when the radioisotope stock bottle (primary vial) is opened. If the material is such that there is a possible pressure build-up during shipment or storage, or if the material has volatile components such as tritium gas or iodine, the container must be opened in a fume hood with an adequate face velocity (>100 lfm < 150 lfm). Open any container with millicurie-level activity in a fume hood. Always assume the outside of the container/vial is contaminated once it is opened, and handle accordingly. - Direct Manipulation
Most inadvertent contamination of laboratory surfaces is caused by contact with contaminated work gloves. Nearly all radioisotope work will involve some direct handling of open containers. Whenever this occurs, assume the gloves are contaminated. Radioiodines and tritium will penetrate disposable gloves, and frequent changing at intervals of not greater than 5 minutes is advised. Also, change gloves immediately if a "clean" item is to be handled. Never wear the gloves away from the immediate work area after direct handling and check them frequently with a survey meter. - Remote Manipulation
For radioisotopes presenting an exposure hazard, or any millicurie quantities, some remote manipulation may be necessary. Use of tongs, forceps, pliers, etc., will lower the radiation dose to the hands and reduce contamination spread via gloves. Metal implements should be rubber tipped for a more secure grip. Whenever an unfamiliar technique is used, practice first with low-hazard materials. Tools are likely to become contaminated and should be frequently checked and cleaned after each use. If equipment is to be used continuously, label and store it separately. EH&S will advise on remote manipulation techniques and equipment. - Transfer
When making liquid transfers, work as far to the rear of the bench as possible in order to avoid personnel and floor contamination from drips, spills, or splattering. Keep equipment at a safe distance from the transfer. For large volumes of radioactive solution, a tray should be used so all the liquid can be contained in case of a spill. Use aids such as pipettes and funnels whenever possible. Cap solutions not to be used immediately.
Pipetting of radioactive materials with any mouth operated system is forbidden by campus regulations. EH&S has product information about hand-operated devices that are compatible with most systems.
When moving a radioisotope solution away from the laboratory bench, secondary containment is necessary. A sealed, plastic bag around the closed primary container is adequate. Rigid, covered unbreakable carriers are preferable, and are required if radioisotopes need to be taken through a corridor.
Self Monitoring
When working with radioisotopes other than 3H, have a portable survey meter on-hand to monitor exposure levels and to check for contamination during and after each experiment. An instrument with an audio response should be used. Call EH&S (2-1493) for advice on survey meter purchases.
In general, to check for equipment or personnel contamination, place the survey meter on the work bench shielded from high background caused by in-process or stored radioactivity. Bench or floor surfaces should be checked by swiping with a piece of filter paper (e.g. Whatman #1), then monitoring the wipe. Care should be taken to avoid contamination of the instrument.
With lower energy beta emitters (3H), swipe surfaces (100 cm2) with a filter paper and count by liquid scintillation. If extensive or high-level surface contamination is suspected, call EH&S. Suggested areas to be examined: floor in front of the work area, reusable equipment, any items handled with work gloves during the experiment (e.g., faucet handles, drawer handles, pipettes, etc.).
Documented monitoring of the radioactive work area should be done at least twice a month if work with radioactivity has occurred.
- When using a survey meter, any surface or article reading greater than background should be considered contaminated. If you are not sure whether the field is produced by a stored source or surface contamination, take a wipe test to rule out removable contamination.
- Any wipe test yielding greater than 60 counts above background per 100 cm2 of surface area is considered an indication of contamination and the area should be decontaminated.
Clean-Up
All items involved in the experiment must be either discarded, stored properly, or surveyed and cleaned. Reusable contaminated glassware must be rinsed at least twice into the liquid radioactive waste before cleaning. There are a number of cleaning compounds that are designed to decontaminate glassware, and adequate cleaning may be accomplished by using strong detergents. Contact EH&S for further information. An overnight soak is usually sufficient but remember to check the activity level of the water before disposing down the sanitary sewer. The equipment may then be cleaned in the following manner:
Rub alternately with a wet, soapy paper towel and a dry one. Discard the towels into the radioactive waste after each application. One sink should be designated for radioisotope clean-up purposes and all glassware and liquids that might be slightly contaminated should be placed there and labeled appropriately.
Storage
Radioactive storage containers and enclosures must be properly labeled. This includes cabinets, refrigerators, cold rooms, etc. Make sure all receptacles are closed tightly and have some secondary containment. Many vessels are permeable in time to certain compounds, especially 3H labeled materials of high specific activity. Such leakage has resulted in several cases of contaminated freezer ice. These compounds should be stored in rigid, secondary containment such as glass. Shield storage areas if necessary so that there is less than 2 mR/hr at contact. Dispose of all unneeded radioisotopes as soon as possible. When a storage enclosure is to be decommissioned, EH&S must check it for contamination and remove the labels.
Airborne Hazards Control
An EH&S, Health Physics approved enclosure must be used whenever there is a possibility of airborne radioactivity. In general, all precautions and procedures mentioned in the preceding sections also apply to fume hood work. Protective devices must be used in conjunction with proper handling procedures, not in place of them. Follow these procedures to use the safety features of a fume hood effectively:
- Call EH&S (530-752-1493) for an air flow test before using the fume hood. Never remove sashes or otherwise alter the fume hood without prior EH&S approval.
- Remove all unnecessary items from the fume hood to prevent their contamination and to maintain the efficiency of the fume hood. Cover stationary objects not to be used.
- Keep radioisotopes away from the sash openings to ensure containment.
- Never enter the sash opening without protection and avoid placing your head inside the plane of the fume hood opening.
- Further protection can be supplied by working around the sash, or doing certain manipulations through a plastic bag in the fume hood. Dry runs are advisable for unfamiliar procedures.
Radiation Accidents
An accident is defined as any unplanned happening that could affect radiation safety. Often the most difficult problem is the recognition that an accident has occurred. The main priorities after an accident are human safety and the protection of the environment.
Radiation Accident Instructions:
- Remove yourself from immediate danger, but do not leave the vicinity.
- Call EH&S at (530) 752-1493 during work hours, or the Police/Fire Department (911) at all other times.
- Mark the spill boundary with tape or other appropriate means.
- Keep all persons out of the accident area until help arrives and do not remove anything from the accident area.
- Persons directly contaminated by a wet spill should remove possibly contaminated clothing and place it in a plastic bag. Any contaminated areas of the body should be washed with mild soap and tepid water.
- If you believe you may have inhaled or ingested radioactive materials, it is most important that you contact EH&S. EH&S and the Occupational Physician will collaborate in developing a suitable bioassay procedure to determine the extent of your exposure and whether any special corrective measures are needed.
Spills
For low level spills (uCi amounts), follow the S.W.I.M. procedures:
Stop the spill - stop the leak or upright the container. Wipe up small spills with absorbent material.
Warn others- Notify personnel in the immediate area and call EH&S.
Isolate the area - Close doors, roope off area, or verbally warn personnel.
Minimize your own exposure to contamination and radiation - move to the edge of the area minimizing spread of contamination or move out side the area and closing the door.
Decontamination Procedures
The basic decontamination sequence is: 1) locate and contain the contamination, 2) assess the contamination and plan clean-up operations, 3) reduce the contamination by appropriate methods, and 4) assess the residual contamination and repeat the procedure if necessary.
- Personnel
Internal contamination will be referred to an appropriate physician if significant. In general, physicians try to eliminate as much of the contamination as possible to prevent or reduce its uptake into the bloodstream and tissues, and to try to prevent fixation of the contaminant or increase its rate of excretion from the body. Vomiting and expectoration may be induced if the contaminant is swallowed or inhaled. In case of small open wounds, cuts, or punctures, or other injuries, the wound should be washed immediately and measured with a survey meter or wipe test. Bleeding may be encouraged. Medical personnel will make these evaluations with the assistance of EH&S and carry out appropriate procedures.
External contamination may be removed by washing with warm water and mild soap. A soft brush may help, but do not abrade the skin. Wash for a few minutes, then dry the area and monitor. Repeat washing if necessary, provided there is no indication of damage. Attempts to remove contamination that resists washing should only be made under medical supervision. - Equipment
Decontamination should be done as soon as possible after use to prevent drying and fixation. Glassware can be cleaned with chromic acid solution or comparable alternative. For other equipment, a 2% solution of isoclean in water is a good general cleaning fluid. Please remember liquid soaking solution should be monitored by liquid scintillation counting or gamma well counting before disposal. If it is greater than the background cpm plus three times the square root of the background cpm, it must be considered radioactive liquid waste.
Do not pour any radioactive material down the drain. Disposal may sometimes be easier and less costly than decontamination. Equipment contaminated with a short-lived radionuclide (e.g., 32P) may be stored behind appropriate shielding until the radionuclide decays to an acceptable level (indistinguishable from background when measured by a survey meter). - Other Surfaces/Areas -Use appropriate survey meter to evaluate contamiation and determine the perimeter of the contamination. Absorb standing liquids and dispose of waste in a radioactive waste container. Clean residual using absorbent paper and soap or decontaminating agent. Begin from the outside of the spill, working your way inward. After decontamination, survey the area again to prove you were successful.
Waste Disposal
All potentially contaminated material must be disposed of as radioactive waste. The radioactive waste will be picked up by a member of the Environmental Health and Safety staff. In order to have a radioactive waste pick up, you must submit a pick-up request online. Waste will be picked up as soon as possible, typically 2-3 working days. Containers must be properly labeled and sealed before they will be picked up. For infrequent users, less than full containers will be accepted for disposal.
Radioactive Waste Disposal Procedures
- Solid Waste
- EH&S provides two cubic-foot cardboard boxes and labels. Clear plastic liners (4 ml and available from Central Storehouse) are required to prevent contamination of the boxes.
- Syringe needles must be placed in hard walled containers in order to prevent puncturing of personnel and the plastic liners.
- No liquid, aerosol, or ether cans are to be placed in solid waste boxes
Vendor shipping containers that have not been contaminated should be disposed of in normal trash cans after the labels and markings are removed or obliterated, and appropriate monitoring done (GM survey meter or wipe test).
- Liquid Waste
- EH&S provides five gallon jerry jugs with labels. No other containers are authorized.
- Only liquid material should be placed in the jerry jugs. No glassware.
- If wash water is less than the background cpm plus three times the square root of the backgound cpm, it can be poured down the sink.
- Segregate liquid waste according to chemical composition described in the EH&S Radioactive Waste Disposal SafetyNet.
- Biological Waste
- Material must be contained in double plastic bags and stored in a freezer until picked up.
- Freezer space availability will dictate the pick-up date by EH&S.
- To make special arrangements for large volumes, please call EH&S before commencing with the experiment.
- Scintilation Vials and Other Small Containers with Liquid Contents
- Keep the scintillation vials separate from other types of waste.
- Scintillation vials should be placed in flats and packed in their original boxes.
- Empty flats may be obtained from EH&S (530-752-1493).
Hazard Evaluations for Commonly Used Radionuclides
-
PHYSICAL DATA
3H
14C
32P
35S
125I
Mode of Decay
Beta
Beta
Beta
Beta
Electron Capture
Primary Radiations
Beta
Max.
18.6
keV
Beta
Max.
156
keV
Beta
Max.
1710
keV
Beta
Max.
167
keV
Gamma
35 keV
X-ray
27 keV
X-ray
31 keV
Half-Life
12.3 years
5730 years
14.28 days
87.4 days
60.1 days
EXTERNAL RADIATION
*
*
*
*
*
Dose Rate/mCi
@ 1 cm
0
160 rad/hour
853 rad/hour
194 rad/hour
1400 mR/hour
Dose Rate/mCi
@ 30 cm
0
< 0.001 rad/hour
0.7 rad/hour
< 0.001 rad/hour
1.6 mR/hour
Beta Range in Air
0.5 cm
21.8 cm
600 cm
24.3 cm
NA
Beta Range in Lucite
5x10-4 cm
2.4x10-2 cm
0.7 cm
2.7x10-2 cm
NA
Preferred Survey Instrumentation
None
Pancake Probe
Pancake Probe
Pancake Probe
Scintillation Probe (NaI)
INTERNAL RADIATION
*
*
*
*
*
Inhalation:
(Target Organ & ALI)
Whole Body
81 mCi
Whole Body
2.4 mCi
Whole Body
0.27 mCi
Whole Body
2.1 mCi
Whole Body
0.54 mCi
Ingestion:
(Target Organ & ALI)
Whole Body
81 mCi
Whole Body
2.4 mCi
Whole Body
0.54 mCi
Whole Body
5.4 mCi
Whole Body
0.27 mCi
Critical Organ
Whole Body
Fatty Tissue
Bone
Bone
Thyroid
Preferred Monitoring Techniques
- 3 H - Liquid scintillation counting of wipes for routine monitoring and locating gross contamination. The low energy beta particles cannot be detected with a GM survey meter.
- 14C - Liquid scintillation counting of wipes for routine monitoring. A "thin-window" preferrably "pancake" probe with a GM survey meter may be used to locate gross contamination.
- 32P - Liquid scintillation counting of wipes for routine monitoring. A "thin-window" preferrably "pancake" probe with a GM survey meter may be used to locate gross contamination and checking the effectiveness of shielding.
- 35S - Liquid scintillation counting of wipes for routine monitoring. A "thin-window" preferrably "pancake" probe with a GM survey meter may be used to locate gross contamination.
- 125I - Liquid scintillation or gamma well counting of wipes for routine monitoring. A scintillation probe is preferred in order to locate gross contamination.
External Radiation Dosimetry
- 3H - Tritium does not present an external hazard because the low energy beta particles cannot penetrate the outer (dead) layer of skin.
- 14C - Carbon-14 does not present an external hazard because the low energy beta particles barely penetrate the outer (dead) layer of skin.
- 35S - Sulfur-35 does not present an external hazard because the low energy beta particles barely penetrate the outer (dead) layer of skin.
Special Considerations
- 3H - Tritiated thymidine is approximately 5 times more radiotoxic than tritiated water under occupational exposure conditions. Tritium migrates through many substances, including surgical/disposable gloves; therefore, keeping surface contamination to a minimum is very important. Wear two pairs of gloves while handling tritiated compounds and change the outer pair frequently. In addition, many tritium compounds are volatile and fume hood use is recommended for procedures involving activities greater than 1 millicurie.
- 14C - Some carbon-14 compounds may penetrate gloves, so wear two pairs of gloves and change the outer pair frequently. Skin contamination can result in significant doses to the skin and underlying tissues.
- 32P Phosphorus-32 is the highest energy beta emitter commonly encountered in the research laboratories and as such requires special handling considerations:
- Shielding - High energy betas emitted by P-32 elicit the production of x-rays when they interact with matter. X-ray production is directly proportional to the atomic mass of the absorber, therefore, shielding with low-Z materials (e.g., plexiglass, lucite) is required. At a minimum use at least 1/4 inch of plexiglass. This thickness of plexiglass will virtually stop all beta emissions from P-32.
- Handling - The dose rates from unshielded sources of P-32 are significant; therefore, the use of plexiglass shielding and remote manipulation of the vials is necessary. Whenever possible, shielding and distance should be maintained during handling procedures and the time of exposure minimized
- 35S - Skin contamination can result in significant doses to the skin and underlying tissue. Be aware that there are reports identifying volatility and contamination issues involving S-35 methionine when the compound's temperature is elevated above room temperature (i.e., incubation of samples).
- 125I - Many I-125 labeled compounds are volatile and certain laboratory procedures release volatile products; therefore, the potential for internal deposition of radioiodine exists. The I-125 concentrates in the thyroid gland and delivers a significant radiation dose over time. You can minimize your potential for internal contamination by:
- Working in a fume hood with an adequate face velocity (i.e., between 100 and 150 lfm). This is required for most I-125 procedures.
- Opening storage containers (e.g., primary vials) in a fume hood.
- Keeping solutions "basic" in order to minimize free iodine release.
- Handling potentially contaminated vials remotely.
- Wearing two pairs of gloves and changing the outer pair frequently.
I-125 emits low energy, penetrating x and gamma ray radiations. Lead foil shielding may be required for experiments involving millicurie activities.
Definitions
Absorbed Dose - The energy imparted by ionizing radiation per unit mass of irradiated material. The units of absorbed dose are the international unit, gray (Gy) or the rad.
Quantity Name Symbol Units Conversions Absorbed Dose gray Gy J/kg 1 Gy = 100 rads rad (old unit) rad 100 ergs/gram 1 cGy = 1 rad Activity - The rate of disintegration per second (dps), minute ( dpm) or decay of radioactive material. The units of activity are the international unit, becquerel (Bq) or the curie (Ci).
Quantity Name Symbol Unit Conversions Activity becquerel Bq dps 1 Ci = 3.7 x 1010 Bq curie (old) Ci 3.7 x 1010 Bq 1 Ci = 3.7 x 1010 Bq Sub units of the curie are:
- millicurie (mCi) = 3.7 x 107 dps
- microcurie (Ci) = 3.7 x 104 dps
- nanocurie (nCi) = 3.7 x 101 dps
- picocurie (pCi) = 3.7 x 10-2 dps
Bioassay - The determination of kinds, quantities or concentrations and in some cases, the locations of radioactive material in the human body, whether by direct measurement (in vivo counting) or by analysis and evaluation of materials excreted or removed from the human body.
Contamination - Deposition of radioactive material in any place where it is not desired.
Contamination Control Zone - A section of a restricted area that has been set up to control the potential spread of contamination from the use of unsealed radioactive materials. A contamination control zone can cover an area as small as a portion of a laboratory bench top or, in rare cases, can encompass an entire room. Contamination control zone guidance is in Section V(D).
Dose Equivalent - The product of the absorbed dose in tissue, quality factor (i.e., rad x Q = rem) or organ dose weighting factors (i.e., Gy x wT = Sv), and all the necessary modifying factors at the location of interest. The units of dose equivalent are the international unit, sievert (Sv) or the rem.
Quantity Name Symbol Unit Conversions Dose Equivalent Sievert Sv J/kg 100 rem = 1 Sv rem (old unit) rem 10-2 Sv 1 rem = 1 cSv Deep Dose Equivalent - External whole body exposure that is the dose equivalent at a tissue depth of one centimeter (1000 mg/cm2).
Exposure - A measure of the ionization produced in air by x or gamma radiation. The sum of electric charges on all ions of one sign produced in air when all electrons liberated by photons in a volume of air are completely stopped in air, divided by the mass of the air in the volume. The units of exposure in air are the international unit, coulomb per kilogram or the roentgen.
Quantity Name Symbol Units Exposure coulomb per kilogram X C/kg roentgen (old unit) R 2.58 x 10-4 C/kg Ionizing Radiation - Any electromagnetic or particulate radiation capable of producing ions directly or indirectly in its passage through matter, In general, it will refer to gamma rays and X-rays, alpha and beta particles, neutrons, protons, high speed electrons, and other nuclear particles. Ionizing radiation does not include radiowaves or visible, infrared or ultra-violet light (i.e., non-ionizing radiation).
Monitoring - Checking for the presence of sources of radiation under a specific set of conditions. Monitoring includes measuring levels of radiation fields and determining contamination levels. Monitoring is performed both for health protection and for protection of future and current research.
Occupational Dose - Occupational dose means the dose received by an individual: In a restricted area; or In the course of employment, education, training or other activities that involved exposure to ionizing radiation.
Personnel Dosimetry - Devices that measure the cumulative dose of radiation to an individual. Types of dosimetry include a film badge, thermoluminescence dosimeter (TLD), TLD finger ring, and albedo-type for neutron measurements.
Radioactive Materials - Any material, solid, liquid or gas that emits ionizing radiation spontaneously.
Roentgen (R) - A unit of exposure to ionizing radiation. It is the amount of gamma rays or X-rays required to produce ions carrying 1 electrostatic unit of electrical charge in 1 cubic centimeter of dry air under standard conditions. See "Exposure."
Roentgen Equivalent Man (rem) - The unit used to express huiman dose equivalence as a result of exposure to ionizing radiation. The relation of the rem to other dose units depends upon the biological effect of the radiation under consideration. For the purposes of this regulation, any of the following is considered to be equivalent to a dose of one rem:
- A dose of one rad due to x, gamma, or beta radiation.
- A dose of 0.1 rad due to neutrons or high energy protons.
- A dose of 0.05 rad due to particles heavier than protons with energy to reach the lens of the eye.
- An exposure of 25 x 106 neutrons per square centimeter, or in accordance with the following table if the energy of the neutrons is known.
Neutron Energy (MeV) Number of Neutrons Per Square Centimeter
Equivalent to a dose of 1 remThermal 980 x 106 0.0001 840 x 106 0.001 980 x 106 0.01 1010 x 106 0.1 170 x 106 0.5 39 x 106 1 27 x 106 2.5 29 x 106 5 23 x 106 7 24 x 106 10 24 x 106 40 14 x 106
Survey Meter - Any portable radiation detection instrument designed to determine the presence of radioactive materials and/or ionizing radiation fields. Survey meters are of two types:
- Count rate meters that detect only the presence of radioactive material. Under certain conditions, the survey meter's reading may be used to determine the exposure rate from a source of radioactive material.
- Dose rate meters that are used to evaluate the intensity of radiation fields in units such as rem per hour, millirem per hour or sievert per hour.
Wipe Test (Sample) - A test (sample) made for the purpose of determining the presence of removable radioactive contamination on a surface. A piece of soft filter paper is wiped over 100 square centimeters of the area to be surveyed and counted for radioactivity with an appropriate instrument.
References
- Slobodien, M. "Radiation Hazards in the Laboratory," in Laboratory Safety: Theory and Practice, Fuscaldo, A.A., Erlick, B.J. and B., eds., Academic Press, New York, 1980.
- Moeller, Dade W., "Occupational and Environmental Radiation Protection," Harvard School of Public Health, (August 1987).
- Bushong, Stewart C.: Radiologic Science for Technologies, Ed. 3, St. Louis, 1984, The C.V. Mosby Co.
- Bushong, Stewart C.: Radiologic Science for Technologies, Ed. 3, St. Louis, 1984, The C.V. Mosby Co.
- Eisenbud, M., Environment, Techology, and Health; Human Ecology in Historical Perspective, New York University Press, New York, NY (1978).
- Muller, H.J., "Radiation and Human Mutation, " Scientific American, Vol. 193, No. 5 (1955).
- "The Effects on Populations of Exposure to Low Levels of Ionizing Radiation, " Advisory Committee on the Biological Effects of Ionizing Radiation, Report No. 3, National Academy Press, Washington, D.C., (1980).
- "Recommendations of the International Commission on Radiological Protection, " Publication 26, International Commission on Radiological Protection, Annals of the ICRP, Vol. 1, No. 3 (1977).
- "Recommendations on Limits for Exposure to Ionizing Radiation, " National Council on Radiation Protection and Measurements, Bethesda, MD (1987).
- "Standards for Protection Against Radiation," Title 10, Part 20, Code of Federal Regulations (1993).
General References
- National Academy of Sciences/National Research Council, "The Effects on Populations of Exposure to Low Levels of Ionizing Radiation," Report of the Advisory Committee on the Biological Effects of Ionizing Radiation, U.S. Government Printing Office, Washington, D.C. (1980).
- "Federal Research on the Biological and Health Effects of Ionizing Radiation," National Academy Press, 2101 Constitution Avenue, N.W., Washington D.C., 20418 (1982).
- National Council on Radiation Protection and Measurements, "Influence of Dose and Its Distribution in Time on Dose-Response Relationships for Low-LET Radiations, NCRP Report No. 64, 1790 Woodmont Avenue, Bethesday MD 20814 (1980).
- National Council on Radiation Protection and Measeurements, "Review of NCRP Radiation Dose Limit for Embryo and Fetus in Occupationally Exposed Women," NCRP Report No. 53, 1790 Woodmont Avenue, Bethesda MD 20814 (1977).
- Brill, A.B. (Editor), Low-Level Radiation Effects: A Fact Book, The Society of Nuclear Medicine, 475 Park Avenue South, New York, NY (1985).